Physics & Science
Calorimetry Explained: The Science of Heat You Never Knew You Were Using Every Day
From boiling water to melting ice — here’s why calorimetry matters more than your textbook ever told you.
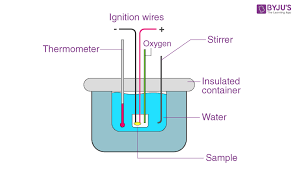
Featured Image: A copper calorimeter with thermometer in a laboratory setting
Here’s something that’ll make you look at your morning coffee differently. The moment you pour hot coffee into a cold mug, calorimetry kicks in. Energy flows from the hot liquid to the cooler ceramic. The coffee cools. The mug warms. And somewhere in between, physics is quietly doing its job — no lab coat required.
I remember sitting in my first physics class, staring at an equation on the board: Q = msΔθ. My teacher said it was “the heart of calorimetry” and I nodded along, pretending I got it. Spoiler: I didn’t. Not until I burnt my hand on a cast iron pan and spent the next five minutes wondering why the pan stayed hot so much longer than the water I’d poured out.
That experience — weirdly enough — was my real introduction to calorimetry. And if you’ve ever asked why some things heat up faster than others, why ice takes so long to melt, or how scientists measure the energy locked inside food… you’ve been asking calorimetry questions without even knowing it.
Let’s dig in — properly this time.
What Is Calorimetry, Really?
At its core, calorimetry is the science of measuring heat — specifically how heat flows between objects, how much is needed to cause a temperature change, and what happens when substances change state (like ice turning to water or water turning to steam).
The word itself comes from the Latin calor, meaning heat, and the Greek metron, meaning measure. So yes — it literally means “heat measurement.” Simple name, surprisingly deep concept.
Here’s the key thing that took me a while to really grasp: heat isn’t something a body “has”. It’s energy in motion. The moment heat stops flowing, it stops being heat — it becomes internal (or thermal) energy of whatever received it. Saying “the heat in my soup” is technically a bit like saying “the running in my legs” — it only makes sense while it’s actively happening.
Heat is the energy transferred between two bodies (or adjacent parts of a body) due to a temperature difference. Once transferred, it becomes the internal energy of the receiving body.
Imagine you’ve just pulled a metal spoon from boiling soup and accidentally touched the handle. That sharp sting? That’s heat transferring from the hot spoon to your skin — fast, unasked-for, and deeply educational. Your hand, being colder, absorbs the energy. The spoon cools slightly. Calorimetry, right there in your kitchen.
— A lesson no textbook teaches quite like experience does
Units of Heat: Joules, Calories, and the Confusion In Between
Here’s where it gets slightly confusing for a lot of students — and honestly, it confused me too. Heat has two widely used units: joules (J) and calories (cal).
The SI unit is the joule, which is consistent with all other energy measurements. But calories have been around longer, and they come up a lot in chemistry, nutrition, and everyday life. You’ve seen “kcal” on food packaging — that’s kilocalories, which is 1000 calories.
The old definition of a calorie was wonderfully specific: the amount of heat needed to raise the temperature of 1 gram of water from 14.5°C to 15.5°C at 1 atm pressure. Scientists eventually nailed down the conversion:
That number — 4.186 — is more important than it looks. It represents one of physics’ great historical breakthroughs: the recognition that heat and mechanical energy are fundamentally the same thing.
Specific Heat Capacity: Why Cast Iron Beats Aluminium in the Kitchen
This is probably the most practically useful concept in all of calorimetry. Specific heat capacity (often just called “specific heat”) tells you how much energy it takes to raise the temperature of 1 gram (or 1 kg) of a material by 1°C.
The formula is:
A higher specific heat means a material needs more energy to heat up — but it also means it holds onto that heat longer. Water has one of the highest specific heat capacities of any common substance (about 4186 J/kg·K), which is why it’s such an incredible coolant and why the ocean moderates coastal climates so effectively.
Here’s a comparison table to make this concrete:
| Material | Specific Heat (cal/g·°C) | Specific Heat (J/kg·K) | Real-life implication |
|---|---|---|---|
| Water | 1.00 | 4186 | Great coolant, used in radiators & cooking |
| Ice | 0.50 | 2093 | Heats up faster than water — but then needs latent heat to melt |
| Aluminium | 0.215 | 900 | Heats & cools fast — ideal for baking sheets |
| Iron | 0.112 | 470 | Retains heat well — cast iron pans stay hot |
| Copper | 0.093 | 389 | Excellent heat conductor — used in calorimeters |
| Lead | 0.031 | 130 | Very low — heats up quickly with small energy input |
So when your cast iron skillet holds heat long after you’ve turned off the stove? That’s specific heat capacity doing its thing. When your thin aluminium baking pan cools down within minutes? Same concept, different number.
Latent Heat: The Hidden Energy Behind Every Phase Change
Alright, here’s where calorimetry gets genuinely fascinating — and where students often get tripped up on exams.
When you heat ice at 0°C, something strange happens: the temperature doesn’t change even as you keep adding energy. You’re not raising the temperature — you’re breaking molecular bonds and converting solid to liquid. This energy is called latent heat (latent meaning “hidden”), and it’s quantified as:
There are two types you need to know:
- Latent heat of fusion: Energy needed to melt a solid (or released when a liquid solidifies). For ice, this is about 3.36 × 10⁵ J/kg.
- Latent heat of vaporization: Energy needed to convert liquid to vapor (or released when vapor condenses). For water, this is approximately 2.25 × 10⁶ J/kg — considerably larger.
Why is vaporization’s latent heat so much larger? Because converting liquid water to steam requires completely separating molecules that still had some attraction in the liquid phase. You’re essentially fighting intermolecular forces across a much larger energy gap.
During a phase change, temperature stays constant — but heat IS still being absorbed or released. Students often confuse “no temperature change” with “no heat transfer.” They’re not the same thing.
The Principle of Calorimetry: The Law That Ties It All Together
Every calorimetry calculation rests on one foundational idea: heat lost by hot objects equals heat gained by cold objects — assuming no heat escapes to the surroundings.
It sounds almost too simple. But this principle, when applied carefully with all the right terms (accounting for latent heat, specific heat of the container, water equivalent, etc.), lets you calculate things that would otherwise seem unknowable — like the specific heat of a completely new material or the latent heat of an exotic substance.
Real-World Example: Mixing Hot and Cold Water
Say you mix 200g of water at 80°C with 300g of water at 20°C. What’s the final temperature? Using the principle:
Heat lost = Heat gained
200 × 4200 × (80 − T) = 300 × 4200 × (T − 20)
Solving gives T = 44°C. Neat, right? No guessing, no approximation — just the physics.
How Scientists Actually Measure Specific Heat: Regnault’s Apparatus
In a lab setting, measuring specific heat precisely requires more care than a kitchen experiment. The classic tool is Regnault’s apparatus — a setup that’s been a staple of physics labs for over a century.
Here’s how it works, roughly:
- A solid object of known mass is heated to a high, measured temperature (using a steam chamber).
- It’s then dropped into a calorimeter containing water at a known temperature.
- The mixture reaches an equilibrium temperature, which is recorded.
- Using the principle of calorimetry, the specific heat of the solid is calculated.
The trickier part is accounting for the heat capacity of the calorimeter itself (usually copper). You can’t just ignore the container — it absorbs heat too. This is why problems often mention “water equivalent of the calorimeter,” which is the mass of water that would have the same heat capacity as the container.
🎓 Beginner’s Guide to Calorimetry Problems
If you’re just starting out, here’s the step-by-step approach that works for almost every calorimetry problem:
List everything you know. Write down all masses, temperatures, specific heats, and latent heats given in the problem. Don’t skip units.
Identify what’s losing heat and what’s gaining heat. The hotter object loses; the colder one gains. Phase changes (melting, boiling) absorb heat.
Check for phase changes. If any substance changes state, you’ll need to include latent heat terms (Q = mL) in addition to sensible heat terms (Q = msΔθ).
Apply the principle: Heat Lost = Heat Gained. Set up the equation carefully, keeping signs consistent.
Check your answer makes physical sense. The final temperature should always be between the initial temperatures of the substances involved (unless a phase change happens at a specific temperature).
The Mechanical Equivalent of Heat: When Physics Got Mind-Blown
Here’s a bit of history that genuinely amazed me when I first learned it. For a long time, scientists didn’t think heat and mechanical energy were the same thing. Heat was a mysterious fluid called “caloric.” Mechanical work was something entirely different.
Then came Joule — and later Searle — who demonstrated experimentally that doing mechanical work on a system raises its temperature in exactly the same way as adding heat. The mechanical equivalent of heat (J = 4.186 J/cal) was measured, and with it, the caloric theory was buried for good.
The Searle’s Cone Method is a beautifully clever experiment: friction between two rotating conical surfaces does work, which heats up a water-filled calorimeter. By measuring the work done and the temperature rise, you can calculate J directly. It’s the kind of elegant experiment that makes you appreciate how much thought went into building the foundations of thermodynamics.
🔥 Pro Tips for Mastering Calorimetry
Students routinely forget that the copper vessel absorbs heat too. The water equivalent of the calorimeter must be added to the mass of water when computing heat gained.
Mixing calories and joules, or grams and kilograms, is the single biggest source of errors. Pick one unit system at the start and don’t switch mid-problem.
In problems involving phase changes, first check whether there’s enough heat for the full phase change to happen. Often, only partial melting occurs — and the final temperature is 0°C, not something you calculate algebraically.
When steam condenses and then cools, there are two separate heat-loss events: the condensation itself (Q = mL) and the cooling of the resulting hot water (Q = msΔθ). Both must be included.
Laboratory answers always differ slightly from theoretical ones because no calorimeter is perfectly insulated. In exam problems, “assuming no heat is lost to surroundings” is your license to ignore this — but in lab reports, always mention it as a source of error.
Common Mistakes Students Make in Calorimetry
Forgetting to include latent heat when there’s a phase change. If ice is melting in your problem, you can’t just use Q = msΔθ for that portion. The melting itself consumes energy at Q = mL.
Using the wrong temperature difference. Δθ should always be (final temp − initial temp) for the object gaining heat, and (initial temp − final temp) for the object losing heat. Getting this backwards flips your signs and wrecks the calculation.
Ignoring the heat capacity of the container. A copper calorimeter with a stirrer has its own heat capacity. In precise problems, this is non-negligible — sometimes it accounts for 10–20% of the total heat gained.
Confusing “specific heat capacity” with “heat capacity.” Specific heat capacity is per unit mass (J/kg·K). Heat capacity is for a particular body (J/K). The heat capacity of an object = its mass × its specific heat capacity.
Assuming the final temperature is always somewhere in the middle. If a phase change is involved, the system might “stall” at 0°C (for ice-water) or 100°C (for water-steam) until the phase change is complete. The final temperature might be exactly these values, not a blend.
Where Calorimetry Shows Up in Real Life
You might think calorimetry is just textbook physics. It’s not. It’s everywhere — and some of its applications are genuinely fascinating.
Food Science and Nutrition
When a food label says a chocolate bar has 250 kcal, that figure was determined using bomb calorimetry — burning the food in a closed chamber and measuring the heat released. The “calorie” in nutrition is actually a kilocalorie (1000 cal), which is one reason nutritionists often write it with a capital C.
Engineering and Material Design
Choosing the right material for heat exchangers, engine components, or building insulation all depends on specific heat capacity values. Engineers designing spacecraft thermal shields, for example, need to know precisely how much energy a material can absorb before its temperature becomes dangerously high.
Climate Science
Water’s extraordinarily high specific heat capacity is one reason Earth’s climate is liveable. Oceans absorb enormous amounts of solar energy without heating up drastically — acting as a massive thermal buffer. Without this property, temperature swings between day and night would be catastrophic.
Medical Applications
Differential scanning calorimetry (DSC) is used in pharmaceutical research to study how drug molecules interact with heat — helping scientists understand stability, purity, and phase behaviour of compounds. It’s a direct descendant of the same principles we’ve been discussing.
Let’s Solve a Real Problem Together
Theory is great, but let’s get practical. Here’s a classic calorimetry problem — the kind you’ll find in competitive exams — solved step by step.
100g of ice at 0°C is placed in 200g of water at 25°C. How much ice melts? (Specific heat of water = 4200 J/kg·K, Latent heat of fusion of ice = 3.4 × 10⁵ J/kg)
Step 1: Calculate heat released by water cooling from 25°C to 0°C.
Q = msΔθ = 0.2 × 4200 × 25 = 21,000 J
Step 2: Calculate heat needed to melt all 100g of ice.
Q_melt = mL = 0.1 × 3.4 × 10⁵ = 34,000 J
Step 3: Compare. Water can only provide 21,000 J, but melting all the ice needs 34,000 J. So only some ice melts.
Step 4: Calculate how much.
m = Q / L = 21,000 / (3.4 × 10⁵) ≈ 62 g
Answer: About 62 grams of ice melts. The final temperature is 0°C (some ice remains unmelted).
See how the principle of calorimetry guides every step? Heat lost by the water = Heat used to melt the ice. Clean, logical, and — once you get the hang of it — almost satisfying to solve.
Frequently Asked Questions About Calorimetry
Q1: What’s the difference between heat and temperature?
Temperature is a measure of the average kinetic energy of molecules in a substance — it tells you how hot something is. Heat is the energy actually being transferred because of a temperature difference. A bathtub of lukewarm water contains more heat energy than a burning matchstick, even though the match is at a higher temperature.
Q2: Why does water have such a high specific heat capacity?
Water molecules form hydrogen bonds with each other. Breaking these bonds (even partially, during heating) requires energy without necessarily raising the kinetic energy enough to show as a temperature increase. This molecular bonding structure is what gives water its unusually high specific heat — and it’s a huge deal for life on Earth.
Q3: What is a calorimeter and why is it made of copper?
A calorimeter is a vessel used to measure heat exchange between substances. Copper is the material of choice because it has a relatively low specific heat capacity (meaning it doesn’t absorb much heat itself), it’s an excellent thermal conductor (so it quickly equilibrates to the temperature of its contents), and it doesn’t react with most substances.
Q4: Why is steam at 100°C more dangerous than water at 100°C?
This is a fantastic calorimetry question! When steam at 100°C touches your skin, it first releases its latent heat of vaporization (about 2260 J/g) as it condenses, and then releases additional heat as the resulting water cools. Water at 100°C only releases the second part. That extra latent heat makes steam burns far more severe than water burns at the same temperature.
Q5: Can calorimetry be used to measure chemical energy?
Absolutely — and this is one of its most important applications. Bomb calorimetry measures the heat released during combustion reactions, which directly gives the energy content (enthalpy of combustion) of fuels, foods, and chemical compounds. It’s the basis for how we know the caloric content of everything from gasoline to granola bars.
Q6: What is the water equivalent of a calorimeter?
It’s a handy simplification. The water equivalent of a calorimeter is the mass of water that would have the exact same heat capacity as the calorimeter (vessel + stirrer). When doing calculations, you can add this value to the actual mass of water and treat the whole thing as one unified water mass — which simplifies the maths considerably.
Wrapping It All Up: Heat, Measure, Understand
Calorimetry isn’t just a chapter in a physics textbook. It’s the framework that explains why your ice pack keeps a drink cold, why coastal cities have milder weather, why steam burns are worse than hot water burns, and why some cooking materials hold heat better than others.
The key ideas to take away:
- Heat is energy in transit — not something an object “contains”
- Specific heat capacity tells you how much energy a material needs per degree of temperature change
- Latent heat is energy absorbed or released during phase changes, with no temperature shift
- The principle of calorimetry — heat lost = heat gained — underpins every calculation in this field
- Real experiments always have heat losses; the principle applies ideally, and lab work teaches you to account for the real world
Whether you’re a student working through exam problems, a curious adult who wants to understand the physics of cooking, or an engineer who needs to pick the right material for a thermal application — understanding calorimetry gives you a genuine edge. It’s one of those areas where the more you understand, the more you see it operating quietly in the world around you.
So next time you put ice in your drink, take a second to appreciate what’s actually happening. That’s not just ice melting — that’s latent heat at work. And now you know exactly what it means.
📚 Further Reading:
Understanding Thermodynamics: A Beginner’s Guide |
The Physics of Phase Changes Explained |
Calorimetry — Britannica
Quantum Computing Explained : How Grover’s Algorithm Achieves a Square Root Speedup

21 Best AI Tools in 2026 That Will Save You Time, Money & Beat Your Competition

NotebookLM Complete Guide : Master Google’s AI Research Tool Like a Pro in 2026
