Physics · Thermodynamics
Kinetic Theory of Gases: A Complete Guide to Molecular Behaviour, Gas Laws and Real-World Applications
Everything you need to understand how tiny invisible molecules explain the behaviour of every gas around you — from your car tyres to the atmosphere above.
1. What Is the Kinetic Theory of Gases?
If you have ever wondered why a balloon expands when you heat it, or why opening a perfume bottle fills an entire room with fragrance within seconds, the answer lies in the kinetic theory of gases. This theory is one of the most elegant achievements of classical physics — it takes the invisible, microscopic world of molecules and uses it to explain everything we observe about gases in everyday life.
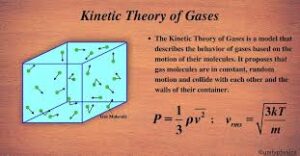
At its core, the kinetic theory says something simple but profound: gases are made of molecules that are always in random motion. These molecules bounce off each other and off the walls of their container constantly. The collective result of billions upon billions of these collisions is what we measure as pressure, temperature and volume. That is all of it. No magic, no mystery — just molecules moving.
Before we go deeper, it helps to understand the concept of an ideal gas. An ideal gas is one that obeys the equation pV = nRT at all conditions. Here, p is pressure, V is volume, n is the number of moles, R is the universal gas constant (8.314 J K⁻¹ mol⁻¹), and T is the absolute temperature. Real gases like oxygen, nitrogen and carbon dioxide behave like ideal gases at low pressure and high temperature, and they deviate when conditions push molecules closer together.
The kinetic theory provides the molecular-level explanation for why this equation works — and why it sometimes does not. Let us go through it piece by piece.
2. The Six Core Assumptions of Kinetic Theory
The kinetic theory is built on a set of assumptions. These assumptions simplify the complicated reality of molecular motion into something we can calculate and predict. It is worth understanding each one because they tell you exactly when the theory works and when it starts to break down.
First, all gases are made of molecules that move randomly in all directions. There is no preferred direction of motion — at any given moment, molecules are flying left, right, up, down, diagonally, and everywhere in between.
Second, the size of a molecule is much smaller than the average distance between molecules. At normal atmospheric pressure and room temperature, the average separation between molecules is roughly 100 times larger than the diameter of a single molecule. This means molecules spend most of their time in empty space, which justifies treating them almost like point particles.
Third, molecules do not exert any force on each other or on the walls of the container, except during a collision. Between collisions, a molecule travels in a perfectly straight line at constant speed. No attraction, no repulsion — just free flight.
Fourth, all collisions are perfectly elastic. This means that kinetic energy is conserved in every collision, whether between two molecules or between a molecule and the wall. No energy is lost as heat or sound. In reality, this holds well at moderate temperatures where molecules do not deform permanently.
Fifth, the molecules obey Newton’s laws of motion. Their movement can be described by classical mechanics — force equals mass times acceleration, and so on.
Sixth, when a gas is left undisturbed for a sufficient time, it reaches a steady state. The number of molecules in any small volume is the same regardless of where you look in the container. The distribution of speeds is also steady — it does not change with time or position.
This steady-state assumption is subtle but powerful. It means that even though individual molecules are colliding and changing velocities all the time, the overall statistical picture stays the same. Because of this, we can effectively ignore individual collisions in our calculations and treat each molecule as if it moves at a constant velocity. This simplification is what makes the pressure calculation tractable.
3. How Pressure Is Derived from Molecular Motion
One of the most satisfying results in all of physics is the derivation of gas pressure purely from the motion of molecules. Let us walk through the logic without getting buried in mathematics.
Imagine a cubical box with side length L. A single molecule moves around inside this box with velocity components vₓ, vᵧ and v_z along the three axes. When this molecule hits one of the walls — say the face perpendicular to the x-axis — the x-component of its velocity reverses (because the collision is elastic), while the y and z components remain unchanged. The change in momentum of the molecule in the x-direction is 2mvₓ, and by Newton’s third law, the wall receives an equal and opposite impulse.
Now, the molecule bounces between the two opposite walls, covering a distance of L in the x-direction each time. The time between two successive collisions with the same wall is 2L/vₓ. So the molecule hits that wall vₓ/(2L) times per second. Each collision delivers a momentum of 2mvₓ to the wall. The force from this one molecule is therefore mvₓ²/L.
Add up the contributions of all N molecules, average over all x-velocity components, and then use the fact that all directions are equivalent (so the average of vₓ² is one-third of the average of v²). The result is:
Here, ρ is the density of the gas and v̄² is the mean square speed (the average of the squares of all molecular speeds). This single equation connects the macroscopic quantity we measure with a pressure gauge directly to the microscopic motion of molecules. It is remarkable that something as simple as bouncing balls in a box gives us the correct formula for gas pressure.
4. RMS Speed and Translational Kinetic Energy
The root mean square speed, or RMS speed, is defined as the square root of the mean square speed. It is written as v_rms and is the most commonly used measure of molecular speed in kinetic theory.
Here M₀ is the molecular weight of the gas. Let us put some numbers to this. Nitrogen at standard conditions (0°C, 1 atm, density 1.25 kg/m³) has an RMS speed of about 490 m/s. That is roughly 1,760 km/h — faster than most aircraft. Oxygen at 300 K has an RMS speed close to 483 m/s. These are not edge cases or extreme conditions; these are the everyday speeds of molecules in the air around you right now.
Lighter molecules move faster. At a given temperature, the average kinetic energy of all molecules is the same, regardless of their mass. So if molecule A is heavier than molecule B, molecule B must be moving faster to have the same kinetic energy. Hydrogen molecules, for instance, move about 3.74 times faster than nitrogen molecules at the same temperature, because nitrogen is 14 times heavier than hydrogen.
The total translational kinetic energy of all the molecules in the gas is:
This is a beautiful result. The total kinetic energy of all the molecules in any ideal gas depends only on the pressure and volume — nothing else. It does not matter what type of gas it is, how many molecules it has, or how heavy those molecules are. Just p and V.
5. Kinetic Interpretation of Temperature
Temperature is one of those concepts we think we understand because we feel it every day. But what is temperature, really, at the molecular level? The kinetic theory gives a precise answer: the absolute temperature of a gas is proportional to the average translational kinetic energy of its molecules.
Here k is the Boltzmann constant (1.38 × 10⁻²³ J/K). This equation is one of the most important in all of physics. It tells us that temperature is nothing more than a measure of how fast molecules are moving on average. A hotter gas has faster molecules; a colder gas has slower molecules.
This also explains what absolute zero means physically. At 0 K (−273.15°C), the average kinetic energy would be zero — meaning molecules would have no translational motion at all. Quantum mechanics adds some nuance here (zero-point energy), but classically, absolute zero is the temperature at which molecular motion stops.
“Temperature is not a thing you pour into a gas. It is a statistical description of how fast the molecules inside it are moving. Heat it up, and you are simply making those molecules run faster.”
An important consequence: when two different gases are mixed and reach thermal equilibrium, their molecules end up with the same average kinetic energy, not the same speed. Heavier molecules will be slower, lighter molecules faster, but the product ½mv² will be the same for all. This principle underlies several fundamental gas laws.
6. Gas Laws Derived from Kinetic Theory
One of the greatest strengths of the kinetic theory is that it does not just explain the ideal gas equation — it derives all the classical gas laws from first principles. Each law emerges naturally as a special case.
Boyle’s Law
At a given temperature, the pressure of a fixed mass of gas is inversely proportional to its volume. Since temperature is constant, v_rms² is constant (because it is proportional to T). The number of molecules and their mass are also fixed. Therefore, from the pressure equation pV = (1/3)Nmv_rms², we get pV = constant, which is exactly Boyle’s law.
Charles’s Law
At constant pressure, volume is proportional to absolute temperature. Since v_rms² is proportional to T, and pV = (1/3)Nmv_rms², at constant p we get V proportional to T. This is Charles’s law.
Avogadro’s Law
Equal volumes of all gases at the same temperature and pressure contain the same number of molecules. Consider two different gases at the same p, V and T. For each gas, pV = (1/3)Nmv². Since they are at the same temperature, the average kinetic energy per molecule is the same for both: ½m₁v₁² = ½m₂v₂². Combining these two relations leads directly to N₁ = N₂.
Graham’s Law of Diffusion
The rate at which a gas diffuses is inversely proportional to the square root of its density. Since v_rms = √(3p/ρ), at the same pressure the ratio of speeds of two gases equals the inverse ratio of the square roots of their densities. Assuming diffusion rate is proportional to speed, this is Graham’s law.
Dalton’s Law of Partial Pressures
The total pressure of a gas mixture equals the sum of the pressures that each individual gas would exert if it were alone in the container. In kinetic theory, the pressure on a wall is just the sum of forces from all individual molecular collisions. Molecules of different gases do not interfere with each other’s contributions. So the total pressure is simply the sum of all partial pressures.
7. The Ideal Gas Equation from Kinetic Theory
Combining the pressure equation with the definition of absolute temperature gives us the full ideal gas equation:
where N is the total number of molecules, k is the Boltzmann constant, n is the number of moles, and R = Nₐk is the universal gas constant. The Boltzmann constant connects the microscopic world (individual molecules) to the macroscopic world (moles of gas), and its value is 1.38 × 10⁻²³ J/K.
This equation lets us calculate a remarkable amount. For example, at 1 atm and 27°C (300 K), one cubic metre of gas contains approximately 2.4 × 10²⁵ molecules. That number is hard to visualise — it is more than two million billion billion molecules per cubic metre. Yet each one is moving at hundreds of metres per second, colliding billions of times per second, and somehow the whole chaotic system produces a smooth, predictable macroscopic pressure that you can read off a gauge.
8. Maxwell’s Speed Distribution Law
The RMS speed is a useful average, but it hides an important truth: not all molecules in a gas move at the same speed. There is a wide spread of speeds, and that distribution has a specific mathematical form known as the Maxwell speed distribution.
Maxwell showed that the number of molecules with speeds between v and v + dv is given by a specific formula involving an exponential term e^(−mv²/2kT). The shape of this distribution has some important features worth understanding.
At any given temperature, most molecules are clustered around a peak speed called the most probable speed (vₚ), but there is a long tail extending to much higher speeds. A small but non-zero fraction of molecules have speeds several times higher than the average. This tail is crucial in chemistry and astrophysics — nuclear fusion reactions in stars, for instance, are driven by the tiny fraction of nuclei with exceptionally high speeds, even though the average temperature might seem too low for fusion.
Notice that the most probable speed is the smallest, the mean speed is slightly larger, and the RMS speed is the largest of the three. All three increase with temperature and decrease with molecular mass. When you heat a gas, the entire distribution shifts to higher speeds, and the peak broadens. When you cool it, everything shifts back toward lower speeds.
The Maxwell distribution also shows why evaporation is a cooling process. Only molecules in the high-speed tail have enough energy to escape the surface of a liquid. When they leave, they carry above-average kinetic energy with them, reducing the average kinetic energy of the remaining liquid — which means a lower temperature.
9. Brownian Motion: Seeing Molecules Indirectly
In 1827, a Scottish botanist named Robert Brown was looking at pollen grains suspended in water through a microscope. He noticed something puzzling: the grains were moving. Not drifting slowly with currents, but jittering randomly — changing direction constantly, moving this way and that with no apparent cause. The water looked perfectly still, and yet the pollen would not stop moving.
Brown had no explanation. He first thought the grains might be alive. He tried it with dead organic material, then with mineral dust. Everything showed the same restless motion. The phenomenon was named Brownian motion, but its cause remained mysterious for decades.
The explanation came in 1905 from Albert Einstein. He showed mathematically that Brownian motion is caused by the uneven bombardment of the particle by water molecules. At any instant, slightly more molecules hit one side of the grain than the other, and the tiny net force kicks the grain in a random direction. The grain is so light that these tiny impacts produce visible movement.
Brownian motion is more intense at higher temperatures (faster molecules, harder kicks) and in less viscous fluids (less resistance to movement). It provided some of the most compelling early evidence that atoms and molecules are real physical objects — not just mathematical conveniences. Einstein’s analysis of Brownian motion even allowed scientists to calculate the size of molecules, something that had been fiercely debated at the time.
10. Vapour, Evaporation and Boiling
The kinetic theory also illuminates the phenomena of evaporation, vapour pressure and boiling — three processes that are closely related but often confused.
Evaporation is a surface phenomenon. Even at temperatures far below the boiling point, some molecules near the surface of a liquid have speeds high enough to overcome the attractive forces of their neighbours and escape into the gas phase. This is why a wet cloth dries even on a cool day, and why perfume evaporates at room temperature. Evaporation is driven by the high-speed tail of the Maxwell distribution.
In a closed container, molecules that escape the liquid will accumulate in the space above it. Eventually, some of them return to the liquid. When the rate of evaporation equals the rate of condensation, equilibrium is reached. The vapour at this point is called saturated vapour, and the pressure it exerts is called the saturation vapour pressure (SVP). The SVP depends only on temperature — it increases as temperature rises, because more molecules have enough energy to escape.
Boiling is different from evaporation. In boiling, molecules throughout the bulk of the liquid gain enough energy to form vapour bubbles, which then rise to the surface. This happens when the SVP of the liquid equals the external pressure acting on its surface. That is why water boils at 100°C at sea level (where atmospheric pressure is 760 mm Hg), but at only 82°C at the top of a high mountain, where atmospheric pressure is lower. A pressure cooker works by raising the pressure inside, which raises the boiling point of water to around 120–140°C, cooking food much faster.
A gas that is below its critical temperature is called a vapour. Above the critical temperature, no amount of compression can liquefy the gas. For water, the critical temperature is 374.1°C. Above this temperature, water exists only as gas, no matter the pressure. Carbon dioxide has a critical temperature of about −56°C (at high pressure), which is why solid CO₂ (dry ice) sublimes directly into gas at atmospheric conditions without passing through a liquid phase.
11. Humidity, Dew Point and Relative Humidity
Water vapour in the atmosphere obeys the same principles as any other gas. Understanding humidity is simply an application of saturation vapour pressure and the Maxwell distribution.
Absolute humidity refers to the actual mass of water vapour present in a given volume of air, typically measured in g/m³. Relative humidity (RH) is the more familiar concept — it tells you how much water vapour is present compared to how much the air could hold at that temperature:
When relative humidity reaches 100%, the air is saturated and cannot hold any more water vapour. Any additional moisture will condense as droplets — forming dew, fog or rain.
The dew point is the temperature at which the current amount of water vapour in the air would saturate it. If the air temperature falls below the dew point, condensation occurs. On a cold morning, the window glass cools the nearby air below its dew point, and tiny water droplets form on the surface — that is dew. When the entire atmosphere in a region cools below the dew point, fog forms as water condenses on dust particles floating in the air.
Relative humidity affects how comfortable or uncomfortable we feel at a given temperature. At high humidity, sweat evaporates more slowly because the air is already nearly saturated with moisture. The body loses less heat, and we feel hotter and stickier. This is why a humid day at 30°C feels far more oppressive than a dry day at 35°C.
Humidity can be measured using a hygrometer. In Regnault’s method, ether is cooled in a silvered test tube until dew appears on the surface. The temperature at this moment is the dew point. Using the SVP tables and the formula above, relative humidity can be calculated precisely.
12. Conclusion: Why the Kinetic Theory Still Matters
The kinetic theory of gases is over 150 years old, and yet it remains one of the most productive frameworks in physics. From a handful of simple assumptions about how molecules move, it generates a complete picture of gas behaviour — pressure, temperature, diffusion, evaporation, vapour pressure, humidity, and more.
More importantly, it changes how you think. Once you internalise the kinetic theory, abstract concepts like temperature and pressure become vivid and concrete. Temperature is not a mysterious quantity — it is just the average speed of molecules. Pressure is not some property of a gas container — it is the accumulated force of billions of tiny molecular impacts every second. Evaporation is not just water disappearing — it is the fastest molecules in a liquid escaping one by one, leaving the slower ones behind and cooling the liquid in the process.
The kinetic theory also has honest limits. Real gases deviate from ideal behaviour when pressures are high or temperatures are low, because molecular size and intermolecular attractions (both ignored in the ideal model) start to matter. The van der Waals equation corrects for these effects. Quantum mechanics introduces further corrections at very low temperatures or for very light molecules like hydrogen and helium. But for most practical purposes — the air in a tyre, the gas in a balloon, the atmosphere above us — the kinetic theory is accurate, elegant, and indispensable.
If there is one takeaway from this entire subject, it is this: the macroscopic world you see and feel is built from the statistical average of an unimaginable number of microscopic events. The temperature on your thermometer is the average speed of 10²⁵ molecules. The pressure in your lungs is the sum of 10²⁵ tiny impacts per second. Everything is motion. Everything is statistics. And somehow, out of all that chaos, emerges perfect, predictable order.
Pressure: p = ⅓ρv̄² |
RMS Speed: v_rms = √(3RT/M₀) |
Avg KE/molecule: ½mv² = 3/2 kT |
Ideal Gas: pV = nRT |
Maxwell Peak: vₚ = √(2kT/m)
How to Start Your Coding Journey in 2026: A Practical Guide for Beginners

Artemis 2 Mission: NASA’s Historic Journey to the Moon